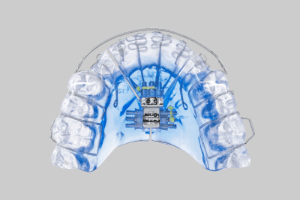
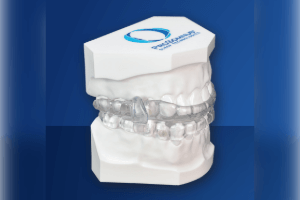
EMA 3D
Summary The EMA 3D is the newest in the family of EMA (Elastic Mandibular Advanacement) devices from EMA-Sleep Inc. All EMA devices use 2 laterally placed elastic straps to advance the mandible and open the bite. The LuxCreo EMA 3D uses on-site 3D printing [...]