Industry Articles
Updates about sleep-related diagnostic devices, therapies, products and companies
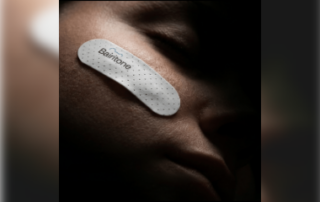
Bairitone Health receives FDA Breakthrough Device Designation for Somnar
Bairitone Health received FDA Breakthrough Device Designation for Somnar, a non-invasive, skin-mounted “sleep sonar” sensor patch paired with an artificial intelligence-driven analysis platform that maps anatomy during sleep to identify the root cause of airway obstruction. About Somnar Somnar is a contact-based system [...]