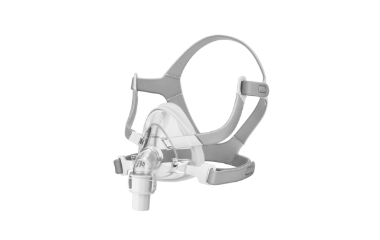
Inogen Inc. has launched a new line of CPAP masks, marking the company’s entry into the sleep apnea market. The company said the Aurora CPAP masks expand its respiratory care portfolio beyond oxygen therapy to include treatment for sleep apnea.
About Aurora CPAP masks
The Inogen Aurora CPAP mask portfolio includes three options: the F1 Full Face mask, the N1 Nasal Cushion mask, and the P1 Nasal Pillows. According to Inogen, the portfolio is intended to address a range of facial structures and therapy needs among patients using CPAP devices. The masks are designed for use in sleep and respiratory therapy and are compatible with most CPAP systems.
All Aurora masks use headgear constructed with a triple-material design and ultrasonic edge sealing technology. The cushions are made of medical-grade rebound silicone and are described as airtight and hypoallergenic. The masks also incorporate a honeycomb vent design intended to reduce noise during use.
The Aurora masks can be assembled, disassembled, and cleaned using standard maintenance steps. The product line has received FDA 510(k) clearance, indicating that it meets applicable safety and performance requirements. Aurora CPAP masks are distributed through certified partners.
The launch of the Aurora masks was supported by a patient-use study conducted in August 2025. Inogen reports that, in this study of 48 patients, a majority of participants indicated high satisfaction with the masks following a 30-day evaluation period.
About obstructive sleep apnea
Obstructive sleep apnea is a sleep disorder in which the airway becomes repeatedly blocked during sleep, causing pauses in breathing. The condition, often associated with loud snoring and choking sounds, can lead to oxygen deprivation and disrupted sleep. Untreated obstructive sleep apnea has been linked to health conditions including high blood pressure, heart disease, stroke, and diabetes.
Read more industry news from the AASM.