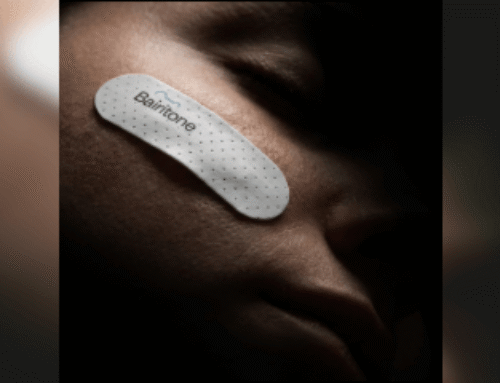
Compumedics announced on Dec. 4, 2023, that its Somfit home sleep test received FDA 510(k) clearance to be marketed in the U.S.
About Somfit
The Somfit system is a non-invasive prescription device intended for home use with patients suspected to have sleep-related breathing disorders. It collects physiological data and is intended to be a diagnostic aid for clinicians.
Somfit is comprised of a single-use adhesive-gel electrode worn on the patient’s forehead, a device that is pressed onto the electrode, an app and a cloud-based data management and reporting system. The Somfit device houses sensors and transmits the data to the app via Bluetooth.
The device calculates and reports to clinicians EEG/EOG channels, sleep stages, oxygen saturation, peripheral arterial tonometry signal, pulse rate and snoring level.
The device previously received regulatory clearance for sale in Australia, New Zealand and the European Union.
About home sleep apnea tests
HSATs are portable devices designed to monitor and record key physiological parameters such as airflow, oxygen levels and breathing patterns during sleep. These convenient diagnostic tools offer an alternative to traditional in-lab sleep studies, providing a more accessible and comfortable option for detecting sleep apnea in the convenience of one’s own home.
Read more industry news from the AASM.